As an emerging class of nanomaterials, nanocrystals (NCs) with few atomic thickness and a lateral size of less than 5nm have recently sparked worldwide interest due to their unique electrical/optical behaviors which are different from the corresponding bulk materials. To date, reports on effective fabrication of ultrafine nanocrystals are still relatively few. Solvent assisted exfoliation and ion insertion are typical traditional preparation method, the former mainly depends on the decreased surface energy of nanoparticles when dispersed in organic solvent through sonication, such as N,N-Dimethylformamide (DMF), N-methylpyrrolidon (NMP), isopropyl alcohol (IPA), and so forth, while the latter mainly focuses on weakening and destroying the van der Waals interactions between the layers of the host materials. Using the above two methods nanocrystals with sizes less than 5 nm can be obtained. However, both of those two methods suffer from multiple drawbacks, including time-consuming/energy-consuming processes, the use of extremely reactive K/Li metal, and tedious battery assembly and high vaccum condition in the glovebox that is not easily handled. Therefore, there is a clear need to explore a simple, general and fast method to prepare nanocrystals under the ambient condition.
The research groups of professor ZHAO Zhigang at Suzhou Institute of Nano-Tech and Nano-Bionics (SINANO), Chinese Academy of Sciences and professor ZHANG Dengsong from Shanghai university launched a detailed and in-depth study on this issue. New progress thus has been made in the preparation of ultrafine monolayered transition-metal dichalcogenide nanocrystals, such as WS2 NCs, MoS2 NCs, WSe2 NCs, and MoS2 NCs. In their design, Al3+ and oleic acid are introduced into the process of electrochemistry and sonication. Trivalent Al3+ ions are employed as intercalating/bombarding ions into the lattice of TMDs, since Al3+ is expected to have a higher charge density and produce much stronger electrostatic forces in the host lattice in comparison with traditional monovalent Li+ ions, which may cause distortion and charge redistribution of the intra-plane covalent bonds, leading to the breaking down of the layer plane of TMDs, just like nails hammered into a plate. Moreover, oleic acid has good affinity for Al3+ and helps to extract Al3+ from intercalated material, which can be favorable to facilitate the efficient formation of small NCs. Finally, monolayered ultrasmall nanocrystals with an average size of 3 nm are obtained. The fast-prepared NCs are then applied to detect highly explosive molecules such as 2,4,6-trinitrophenol (TNP) with a low detection limit of 10-6 M.
Based on prior work, the research groups of professor ZHAO Zhigang, professor ZHANG Ting at SINANO, and professor Geng Fengxia from Soochow University worked together to apply this preparation method in the fabrication of hexgonal boron nitride nanocrystals (h-BN NCs) and cubic boron nitride nanocrystals (BN NCs). They not only succeed in the preparation of two kinds of BN NCs, but also observed their distinct performances on piezoelectricity. Compared to the corresponding bulk materials, both of h-BN NCs and c-BN NCs have a better piezoelectric behavior, The piezoelctric coefficient (d33) of c-BN NCs is up to 25.7 pC/N, which is almost 6 times larger than that from bulk c-BN. Moreover, the piezoelectric coefficient of c-BN NCs is also better than that of h-BN NCs, which may be due to the fast piezoelectric response determined by the cubic structure. This work paves an interesting route for exploring new properties of superhard NCs and has been published in the journal of Nano letters.
The series of work was supported by the National Natural Science Foundation of China, the Outstanding Youth Fund of Jiangsu Province.
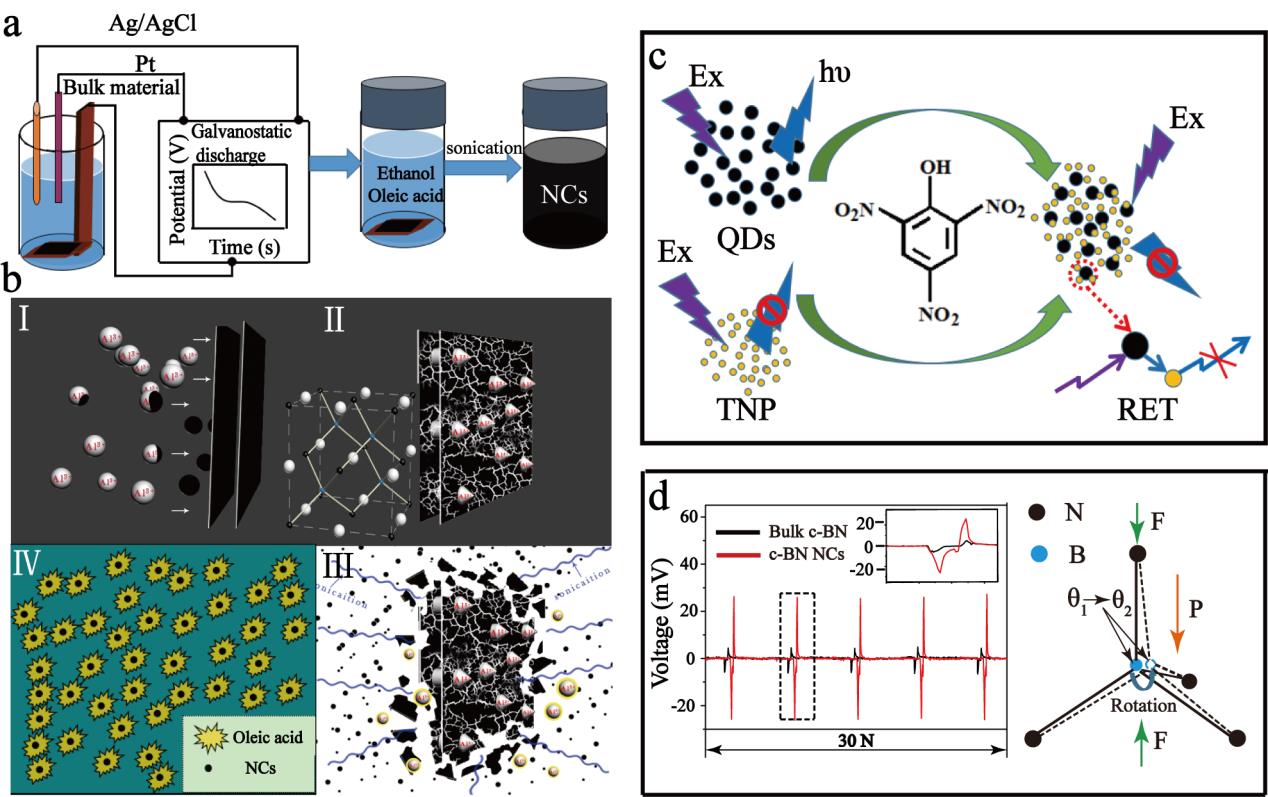
(a) Schematic representation of electrochemical shock process for the fabrication of ultrafine c-BN NCs from the bulk materials. (b) Formation mechanism of c-BN NCs synthesized via
electrochemical shock. (c) Schematic of the proposed mechanism for fluorescence quenching. (d) Output voltage for bulk c-BN and c-BN NCs under the same stress of 30 N (left), the Distortion of the unit cell of c-BN under the compression of an external force, showing the bond angle change (right).(Image by SINANO)
Reference:
Fast preparation of ultrafine monolayered transition-metal dichalcogenide quantum dots using electrochemical shock for explosive detection.
Rapid Synthesis of Sub-5 nm Sized Cubic Boron Nitride Nanocrystals with High-Piezoelectric Behavior via Electrochemical Shock.
Contact Information:
Prof.ZHAO Zhigang
Suzhou Institute of Nano-Tech and Nano-Bionics,Chinese Academy of Science
Suzhou, Jiangsu 215123, China.
Email: zgzhao2011@sinano.ac.cn

