In this new work, C/S composite particles are imperfectly coated with a wrapping layer, and the material is assembled into coin cells. The imperfect wrapping layer allows the infiltration of adequate electrolyte into the C/S composite particles. Significantly, a special functional additive is added to the electrolyte to react with the initially imperfect wrapping layer to form a second wrapping layer after the battery is assembled. The second, post-assembled and in situ-formed wrapping layer is designed to be compact and tight to completely block the shuttle effect, which allows polysulfide dissolution into the electrolyte within the interior of the wrapped composite particles (Figure. 1d).
This in situ wrapping approach solves this trade-off by first allowing the electrolyte to infuse into the interior of the porous cathode material and then closing the pores by a surface reaction with a special additive in the electrolyte. The research results show greatly improved Coulombic efficiency and cycle life of lithium sulfur batteries by this in situ wrapping approach. Importantly, the capacity decay of the cell at 1000 cycles is as small as 0.030% per cycle at 1C. This work has published in Nature Communications.
This research work is supported by the “Strategic Priority Research Program” of the CAS, the Ministry of Science and Technology of China, and Natural Science Foundation of China.

Fig.1. Schematic illustration of the unique in situ wrapping strategy for Li-S cathode structures. (a) The no wrapping case, exhibiting severe capacity decay in cycling. (b) Perfect wrapping of C/S materials before battery assembly, exhibiting poor coverall performance due to lack of electrolyte in the active material. (c) Imperfect wrapping of the cathode material before battery assembly, exhibiting improved cycle stability than the no wrapping case. (d) Perfect post-assembly in situ wrapping of the cathode material, exhibiting ideal cycle stability by blocking polysulfide shuttle while allowing for electrolyte infiltration in the active material.(Image by SINANO)
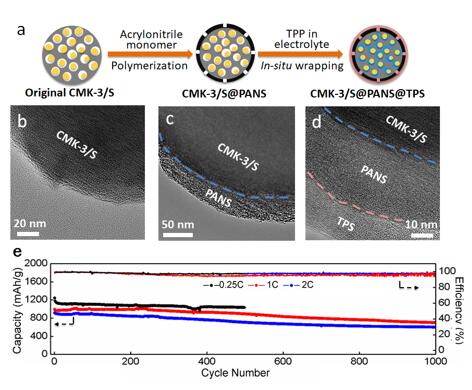
Fig. 2. (a) Schematic illustration of the in situ wrapping process. TEM images of (b) CMK-3/S, (c) CMK-3/S@PANS and (d) CMK-3/S@PANS@TPS particles. (e) Long cycling performance of cells with CMK-3/S@PANS@TPS cathodes at 0.25C, 1C and 2C.(Image by SINANO)
Reference:
https://www.nature.com/articles/s41467-017-00656-8
Contact Information:
Prof. CHEN Liwei, Suzhou Institute of Nano-Tech and Nano-Bionics, Chinese Academy of Sciences
Email: lwchen2008@sinano.ac.cn

